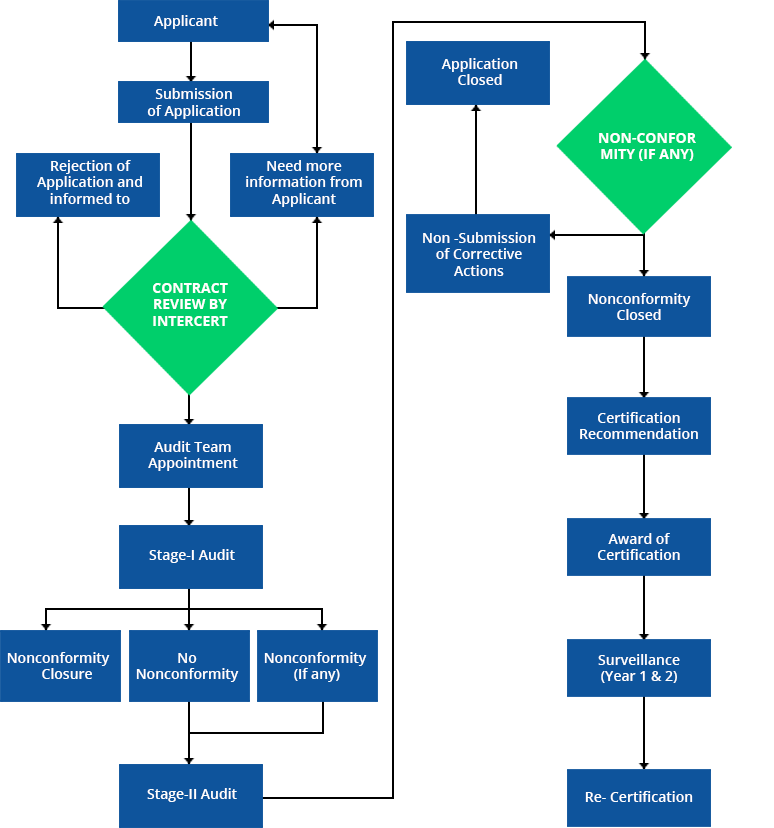
ISO 13485 is designed to be used by organizations involved in the design, production, installation and servicing of medical devices and related services. A medical device is a product, such as an instrument, machine, implant or in vitro reagent, that is intended for use in the diagnosis, prevention and treatment of diseases or other medical conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
